(SC Quantum is an IYQ sponsor.)
When Clemson University students signed up for the first-ever South Carolina Quantathon in October 2024, they didn’t expect it would launch them onto a yearlong journey that would carry them across the globe and to some of the world’s most respected quantum hackathons. But that’s exactly what happened.
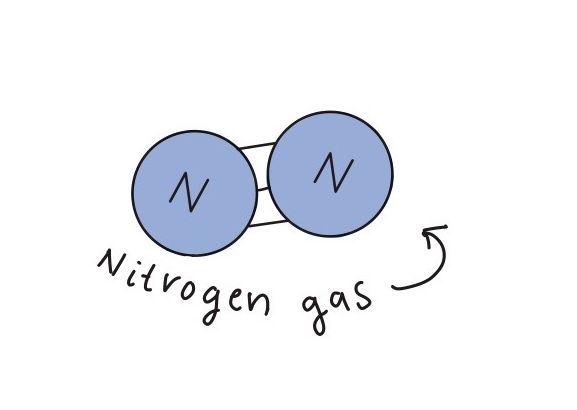
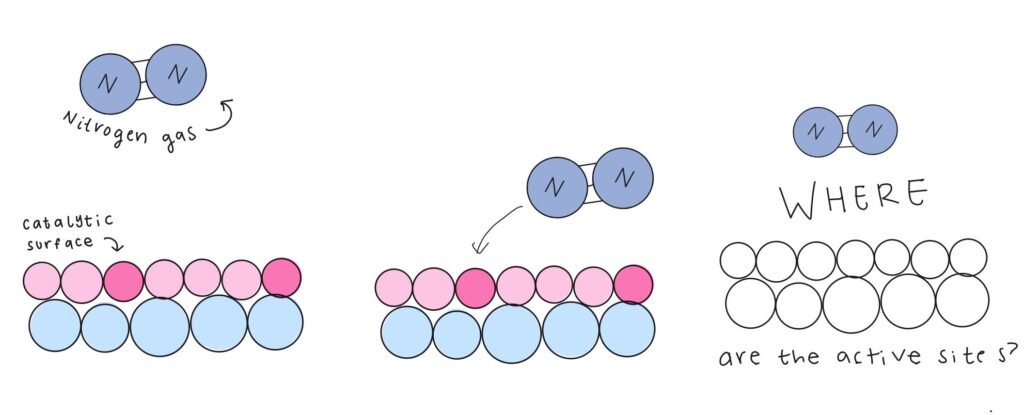
What began as a weekend experience in Columbia, South Carolina, where students worked on quantum random number generation (QRNG) at SC Quantum’s flagship hackathon, has grown into something much larger. Guided by Dr. Rong Ge from Clemson University and supported by cross-sector partnerships, these students now show what can happen when curiosity meets opportunity in the quantum space.
“The original Quantathon catapulted me into a deeper commitment to quantum computing research. It helped me find what I’m passionate about.”
—Valentine Mohaugen, Clemson undergraduate
Much of this momentum was made possible by Clemson’s Creative Inquiry (CI) Program, which supports Dr. Ge’s “Hands-on Quantum Computing” course. Among its broad assistance, CI helps facilitate travel and training opportunities for students who are immersed in this emerging technology.
CI provided critical early support for the team’s participation in the SC Quantathon and MIT iQuHack, experiences that helped launch their year-long journey. Students backed by CI were also part of the team that competed in Abu Dhabi, a major accomplishment that highlights the lasting impact of this investment. The students affiliated with CI include:
- Nathan Jones (PhD CI Mentor)
- Joseph Benich (School of Computing undergraduate, Junior)
- Toby Cox (School of Computing undergraduate, Junior)
- Valentine Mohaugen (Physics, Senior)
- Ian Lewis (School of Computing undergraduate, Junior)
The broad backgrounds, perspectives, and expertise apparent in the team are a dynamic reflection of Clemson and of the interest quantum technology is gaining across campuses across our region.
From First Hack to First Place
At Quantathon V1, the Clemson team took on the quantum random number generation (QRNG) challenge presented by DoraHacks, creating a framework for scalable quantum random number generation and post-processing for cybersecurity and algorithmic applications. They not only won their challenge but also earned the event’s top prize. In partnership with SC Quantum and NYU Abu Dhabi, the team secured spots at the NYU-AD International Hackathon for Social Good in April 2025 in Abu Dhabi. Their win also came with a mini-grant from DoraHacks to continue developing their project, setting off a wave of insight and new opportunities.
“We dug into the tradeoffs between randomness, speed, and efficiency—things we only scratched the surface of during the hackathon.”
—Sam Quan, Clemson undergraduate
Over the following weeks, the team expanded on their original work, refining entropy analysis, comparing algorithmic tradeoffs, and drafting a technical write-up of their findings for DoraHacks. In doing so, they gained new skills in quantum algorithm design, optimization, and applied research.
Learning at MIT and Arriving in Abu Dhabi
The momentum from Quantathon V1 soon carried the team to MIT’s competitive iQuHack. The event brought deep dives into theoretical challenges hosted by Alice & Bob, an elite group of participants, and a fast-paced environment that pushed their understanding to new levels.
The NYUAD experience brought a new kind of learning: large, diverse teams working on open-ended problems with real-world applications. As student mentors, the Clemson group played a leadership role. The experience combined cultural exchange with technical leadership, as they guided group projects in areas like quantum sensing, machine learning, and mental health diagnostics.
“The NYUAD hackathon was the best academic and cultural experience of my life. I made friends I’m going back to visit.”
—Valentine Mohaugen
Their NYUAD projects are now being prepared for publication, a reflection of the depth of their exploration and the professional-level collaboration they achieved through these opportunities.
A New Chapter, and More to Come
At home on campus, the students have helped grow Clemson’s emerging quantum community, supporting peers through a student-led quantum club and mentoring new students entering the space. Today, they’re exploring publications, refining their projects, and even helping plan future SC Quantum events.
Their journey is a vivid example of how faculty support, early access, and cross-sector collaboration can empower undergraduates to thrive in advanced, interdisciplinary fields. The team credits Dr. Ge’s mentorship and Clemson’s academic environment as foundational to their success.

“I’m incredibly grateful to SC Quantum, DoraHacks, MIT, and NYU Abu Dhabi for opening doors for us. But none of it would have happened without Dr. Ge’s support back at Clemson.”
—Sam Quan
SC Quantum’s hackathon model and network of partners played a key role in shaping the team’s path, offering early exposure, funding support, and global visibility. As South Carolina’s regional quantum network expands, stories like this show the value of building pathways that meet students where they are and help them reach even further.
And the best part? This is still the beginning.
– – – – – –
Quantathon V2, the second edition of SC Quantum’s flagship hackathon for the Southeast, will take place October 9–12 in Columbia, South Carolina, at the Darla Moore School of Business at the University of South Carolina. The event is powered by qBraid, with Platinum Sponsors the Columbia Area Development Partnership and AgFirst Farm Credit Bank. Learn more about Quantathon V2 by clicking here.
Dave Alsobrooks
Director of Communications, SC Quantum
dalsobrooks@scquantum.org